Presbyopia Market to Witness Accelerated Growth at a CAGR of 4.9% During the Forecast Period (2026–2036) Supported by Expanding Treatment Landscape | DelveInsight
New York, USA, May 11, 2026 (GLOBE NEWSWIRE) -- Presbyopia Market to Witness Accelerated Growth at a CAGR of 4.9% During the Forecast Period (2026–2036) Supported by Expanding Treatment Landscape | DelveInsight
The presbyopia market is witnessing steady growth driven by the rising global aging population and increasing prevalence of age-related near vision impairment. Presbyopia market expansion is further supported by growing awareness and higher diagnosis rates. Additionally, the launch of emerging therapies and devices such as Phentolamine (Opus Genetics/Viatris), MicroLine (Pilocarpine Ophthalmic) (Eyenovia), GLK-302 (Glaukos Corporation), CorVision (Intrastromal corneal inlay) (LinkoCare Life Sciences AB), riboflavin/UV-A medical device (RegenSight), and others will further boost the market growth.
Recently published Presbyopia Market Insights report includes a comprehensive understanding of current treatment practices, presbyopia emerging drugs, market share of individual therapies, and current and forecasted market size from 2022 to 2036, segmented into leading markets [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
Presbyopia Market Summary
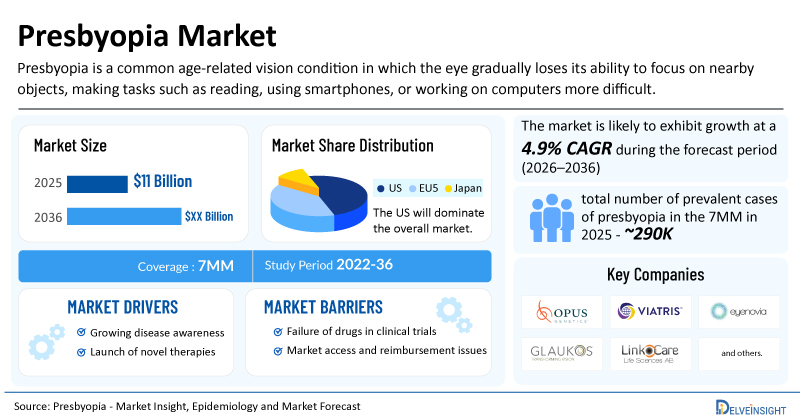
- The market size for presbyopia was found to be USD 11 billion in the 7MM in 2025.
- The United States accounted for the largest presbyopia treatment market size in 2025, i.e 50%, compared to other major markets, including the EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- The total number of prevalent cases of presbyopia in the 7MM was nearly 290,000 cases in 2025 and is projected to increase during the forecasted period.
- Key presbyopia companies, including Opus Genetics, Viatris, Eyenovia, Glaukos Corporation, LinkoCare Life Sciences AB, RegenSight, and others, are actively working on innovative presbyopia drugs.
- Some of the key presbyopia therapies and devices in clinical trials include Phentolamine, MicroLine (Pilocarpine Ophthalmic), GLK-302, CorVision (Intrastromal corneal inlay), riboflavin/UV-A medical device, and others. These novel presbyopia therapies and devices are anticipated to enter the presbyopia market in the forecast period and are expected to change the market.
- Microline (Eyenovia) is the potential topical solution in the development of Presbyopia, with its expected approval by 2027 in the US.
Discover what is the future market of presbyopia treatment @ https://www.delveinsight.com/sample-request/presbyopia-market
Key Factors Driving the Growth of the Presbyopia Market
- Rising Presbyopia Prevalence: In 2025, the US accounted for approximately 125,000 diagnosed prevalent cases of Presbyopia. These cases are expected to increase during the forecast period (2026−2036) owing to increasing populations, improved diagnostic methods, advancements in medical technology allowing for better treatment, and the emergence of gene therapy and next-generation treatments, which could redefine the landscape.
- Rising Aging Population: One of the strongest contributors to market growth is the increasing global elderly population. Since presbyopia is an age-related condition caused by reduced lens flexibility, the growing number of adults aged 40 years and above is directly increasing the patient pool.
- Technological Advancements in Vision Correction: Continuous innovation in multifocal lenses, laser-assisted procedures, corneal inlays, and presbyopia-correcting IOLs has improved safety, precision, and patient outcomes.
- Launch of Emerging Drugs and Devices: The dynamics of the presbyopia market are expected to change in the coming years due to the launch of emerging therapies and devices such as Phentolamine (Opus Genetics/Viatris), MicroLine (Pilocarpine Ophthalmic) (Eyenovia), GLK-302 (Glaukos Corporation), CorVision (Intrastromal corneal inlay) (LinkoCare Life Sciences AB), riboflavin/UV-A medical device (RegenSight), and others.
Stuti Mahajan, consulting manager at DelveInsight, said that Regulatory approvals of pharmacological presbyopia treatments such as VUITY, QLOSI, VIZZ, and YUVEZZI reflect growing clinical validation and regulatory confidence in non-invasive therapeutic options.
Presbyopia Market Analysis
- Management strategies for presbyopia include corrective eyewear such as glasses and contact lenses, along with surgical options like refractive procedures and lens implants specifically intended to restore near vision.
- Multiple surgical techniques are currently available, and continuous technological progress has enhanced both their safety profiles and clinical outcomes. Frequently used refractive surgeries include Conductive Keratoplasty, LASIK, LASEK, and Photorefractive Keratectomy (PRK).
- At present, approved pharmacological treatments for presbyopia in the US include VUITY (AbbVie) and QLOSI.
- Across the seven major markets, the US accounts for the largest market share, with four approved therapies: VUITY, QLOSI, VIZZ, and YUVEZZI. In contrast, Japan and the EU4 + United Kingdom currently have no approved branded therapies for presbyopia, excluding generics and biosimilars.
- Several companies are actively advancing innovative treatments with novel mechanisms of action to address persistent unmet needs and improve patient outcomes.
- However, the late-stage development pipeline for presbyopia remains relatively limited. Promising emerging candidates such as Phentolamine and MicroLine are anticipated to enter the market during the forecast period.
Learn more about the new drugs for presbyopia @ Presbyopia Treatment Market
Presbyopia Competitive Landscape
Some of the presbyopia drugs and devices under development include Phentolamine (Opus Genetics/Viatris), MicroLine (Pilocarpine Ophthalmic) (Eyenovia), GLK-302 (Glaukos Corporation), CorVision (Intrastromal corneal inlay) (LinkoCare Life Sciences AB), riboflavin/UV-A medical device (RegenSight), and others.
Opus Genetics’ Phentolamine Ophthalmic Solution 0.75% is an advanced investigational treatment for presbyopia that functions as a non-selective alpha-adrenergic antagonist. By reducing iris dilator muscle activity and adjusting pupil size, it improves near vision without directly affecting the ciliary muscle. The therapy is presently under regulatory assessment after the submission of a supplemental NDA in early 2026.
A key differentiator of this candidate is its non-miotic mode of action, which may deliver reliable near-vision benefits without negatively affecting distance vision, a drawback often seen with currently available therapies. Backed by encouraging Phase III clinical data, it demonstrates a favorable safety profile and user-friendly dosing schedule, making it a promising next-generation pharmacologic option in the evolving presbyopia market. Opus Genetics expects a regulatory outcome by the end of 2026.
Linkcore’s CorVision is an innovative bioengineered corneal microlens intended for presbyopia correction. Constructed from collagen, the same natural substance found in the human cornea—it is implanted into a corneal pocket, allowing smooth integration while reducing the risks commonly linked to conventional corneal inlays and more invasive surgical procedures.
The anticipated launch of these emerging presbyopia therapies are poised to transform the presbyopia market landscape in the coming years. As these cutting-edge presbyopia therapies continue to mature and gain regulatory approval, they are expected to reshape the presbyopia market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about the top companies in presbyopia market, visit @ Presbyopia Medication
Recent Developments in the Presbyopia Market
- In January 2026, Tenpoint Therapeutics announced it had secured USD 235 million through the successful close of its Series B preferred stock financing and a credit facility. The financing will help in the commercialization of YUVEZZ.
- In September 2025, LENZ Therapeutics, Inc. announced that VIZZ (aceclidine ophthalmic solution) 1.44%, the first and only FDA-approved aceclidine-based eye drop for the treatment of presbyopia in adults, is now available. Professional product sample distribution by the sales force to optometrists and ophthalmologists has been initiated nationwide.
- In July 2025, LENZ Therapeutics, Inc. announced that the US Food and Drug Administration approved VIZZ (aceclidine ophthalmic solution) 1.44%, the first and only FDA-approved aceclidine-based eye drop for the treatment of presbyopia in adults.
What is Presbyopia?
Presbyopia is a common age-related vision condition in which the eye gradually loses its ability to focus on nearby objects, making tasks such as reading, using smartphones, or working on computers more difficult. It typically begins to develop in adults over the age of 40 as the natural lens inside the eye becomes less flexible and the surrounding muscles lose some focusing efficiency. People with presbyopia often experience symptoms such as blurred near vision, eye strain, headaches, and the need to hold reading materials farther away. Although presbyopia is a natural part of aging and not a disease, it can significantly impact daily activities and quality of life. It can be effectively managed with reading glasses, bifocals, contact lenses, or newer prescription eye drops and surgical treatment options.
Presbyopia Epidemiology Segmentation
The presbyopia epidemiology section provides insights into the historical and current presbyopia patient pool and forecasted trends for the leading markets. In the 7MM, the US accounted for the highest number of diagnosed prevalent cases of Presbyopia, with nearly 125,000 cases in 2025.
The presbyopia market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Total Prevalent Cases of Presbyopia
- Total Diagnosed Cases of Presbyopia
- Age-specific Cases of Presbyopia
- Gender-specific Cases of Presbyopia
- Severity-specific Cases of Presbyopia
- Total Treated Cases of Presbyopia
Download the report to understand presbyopia unmet needs in current treatment @ Presbyopia Treatment Options
| Presbyopia Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Presbyopia Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Presbyopia Market CAGR | 4.9% |
| Presbyopia Market Size in 2025 | USD 11 Billion |
| Key Presbyopia Companies | Opus Genetics, Viatris, Eyenovia, Glaukos Corporation, LinkoCare Life Sciences AB, RegenSight, AbbVie, Orasis Pharmaceuticals, LENZ Therapeutics, Tenpoint Therapeutics, and others |
| Key Presbyopia Therapies and Devices | Phentolamine, MicroLine (Pilocarpine Ophthalmic), GLK-302, CorVision (Intrastromal corneal inlay), riboflavin/UV-A medical device, VUITY, QLOSI, VIZZ, YUVEZZI, and others |
Scope of the Presbyopia Market Report
- Presbyopia Therapeutic Assessment: Presbyopia current marketed and emerging therapies
- Presbyopia Market Dynamics: Conjoint Analysis of Emerging Presbyopia Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Presbyopia Market Unmet Needs, KOL’s views, Analyst’s views, Presbyopia Market Access and Reimbursement
Discover more about the clinical pipeline for presbyopia in 2026 @ Presbyopia Clinical Trials
Table of Contents
| 1 | Presbyopia Market Key Insights |
| 2 | Presbyopia Market Report Introduction |
| 3 | Executive Summary |
| 4 | Key Events |
| 5 | Epidemiology and Market Forecast Methodology |
| 6 | Presbyopia Market Overview at a Glance |
| 6.1 | Market Share (%) Distribution of Presbyopia by Therapies in 2025 |
| 6.2 | Market Share (%) Distribution of Presbyopia by Therapies in 2036 |
| 7 | Presbyopia: Overview |
| 7.1 | Introduction |
| 7.2 | Signs and Symptoms |
| 7.3 | Pathophysiology |
| 7.4 | Etiology |
| 7.5 | Diagnosis |
| 7.6 | Treatment & Management |
| 8 | Guidelines of Presbyopia |
| 9 | Regulation of Medical Devices |
| 9.1 | United States Regulation of Devices |
| 9.2 | European Regulation of Devices |
| 9.3 | Japan Regulation of Devices |
| 10 | Epidemiology and Patient Population |
| 10.1 | Key Findings |
| 10.2 | Assumptions and Rationale |
| 10.3 | Epidemiology: 7MM |
| 10.3.1 | Total Prevalent Cases of Presbyopia in the 7MM |
| 10.3.2 | Total Diagnosed Cases of Presbyopia in the 7MM |
| 10.3.3 | Total Age-specific Cases of Presbyopia in the 7MM |
| 10.3.4 | Gender-specific Cases of Presbyopia in the 7MM |
| 10.3.5 | Severity-specific Cases of Presbyopia in the 7MM |
| 10.3.6 | Total Treated Cases of Presbyopia in the 7MM |
| 10.4 | The United States |
| 10.4.1 | Total Prevalent Cases of Presbyopia in the United States |
| 10.4.2 | Total Diagnosed Cases of Presbyopia in the United States |
| 10.4.3 | Total Age-specific Cases of Presbyopia in the United States |
| 10.4.4 | Gender-specific Cases of Presbyopia in the United States |
| 10.4.5 | Severity-specific Cases of Presbyopia in the United States |
| 10.4.6 | Total Treated Cases of Presbyopia in the United States |
| 10.5 | EU4 and the UK |
| 10.6 | Japan |
| 11 | Patient Journey |
| 12 | Marketed Presbyopia Drugs |
| 12.1 | Key Cross Competition |
| 12.2 | VUITY (Pilocarpine HCI): AbbVie |
| 12.2.1 | Drug Description |
| 12.2.2 | Regulatory milestones |
| 12.2.3 | Other Development Activities |
| 12.2.4 | Summary of Pivotal Trial |
| 12.2.5 | Clinical Development |
| 12.2.5.1 | Clinical trials information |
| 12.2.6 | Safety and Efficacy |
| 12.2.7 | Analyst Views |
| 12.3 | QLOSI (Pilocarpine hydrochloride): Orasis Pharmaceuticals |
| 12.4 | VIZZ (Aceclidine): LENZ Therapeutics |
| 12.5 | YUVEZZI (carbachol and brimonidine tartrate): Tenpoint Therapeutics |
| 13 | Emerging Presbyopia Drugs |
| 13.1 | Key Cross Competition |
| 13.2 | Phentolamine: Opus Genetics/Viatris |
| 13.2.1 | Product Description |
| 13.2.2 | Other Development Activities |
| 13.2.3 | Clinical Development |
| 13.2.3.1 | Clinical Trials Information |
| 13.2.4 | Safety and efficacy |
| 13.2.5 | Analyst Views |
| 13.3 | MicroLine (Pilocarpine Ophthalmic): Eyenovia |
| 13.4 | GLK-302: Glaukos Corporation |
| 14 | Marketed Presbyopia Medical Devices |
| 15 | Emerging Presbyopia Medical Devices |
| 15.1 | Key Cross Competition |
| 15.2 | CorVision® (Intrastromal corneal inlay): LinkoCare Life Sciences AB |
| 15.2.1 | Product Description |
| 15.2.2 | Regulatory Milestones |
| 15.2.3 | Clinical Development |
| 15.2.3.1 | Clinical Trial Information |
| 15.2.4 | Analyst Views |
| 15.3 | riboflavin/UV-A medical device: RegenSight |
| 16 | Presbyopia Market: Seven Major Market Analysis |
| 16.1 | Key Findings |
| 16.2 | Market Presbyopia Outlook |
| 16.3 | Conjoint Analysis |
| 16.4 | Key Presbyopia Market Forecast Assumptions |
| 16.5 | The 7MM Presbyopia Market Size |
| 16.5.1 | Total Market Size of Presbyopia in the 7MM |
| 16.5.2 | Total Market Size of Presbyopia by therapies in the 7MM |
| 16.6 | The United States Presbyopia Market |
| 16.6.1 | Total Market Size of Presbyopia in the United States |
| 16.6.2 | Total Market Size of Presbyopia by Therapies in the United States |
| 16.7 | EU4 and the UK Presbyopia Market |
| 16.8 | Japan Presbyopia Market |
| 17 | Presbyopia Market Unmet Needs |
| 18 | Presbyopia Market SWOT Analysis |
| 19 | KOL Views on Presbyopia |
| 20 | Presbyopia Market Access and Reimbursement |
| 20.1 | The United States |
| 20.2 | EU4 and the UK |
| 20.3 | Japan |
| 20.4 | Summary and Comparison of Market Access and Pricing Policy Developments in 2025 |
| 20.5 | Market Access and Reimbursement of Presbyopia Therapies |
| 21 | Bibliography |
Related Reports
Presbyopia Clinical Trial Analysis Pipeline
Presbyopia Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key presbyopia companies, including Orasis Pharmaceuticals, Novartis, Cellix Bio, Visus Therapeutics, AbbVie, Vyluma, Lenz Therapeutics, Ocuphire Pharma, JIXING Pharmaceuticals, Eyenovia, and others.
Myopia Market Insight, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key myopia companies such as Vyluma, Inc., Sydnexis, Inc., Ocumension Limited, Santen Pharmaceutical, Cloudbreak Therapeutics, Nevakar, Inc., Eyenovia, Stuart Therapeutics, Cellix Bio, JeniVision, Zhaoke Ophthalmology, and others.
Myopia Clinical Trial Analysis Pipeline
Myopia Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key myopia companies, including Vyluma, Inc., Sydnexis, Inc., Ocumension Limited, Santen Pharmaceutical, Cloudbreak Therapeutics, Nevakar, Inc., Eyenovia, Stuart Therapeutics, Cellix Bio, JeniVision, Zhaoke Ophthalmology, and others.
Contact Lenses Market Insights, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the market trends, market drivers, market barriers, and key contact lenses companies, including Alcon Inc, Cooper Vision Inc, Johnson & Johnson Vision, Bausch Health Companies Inc., HOYA Vision Care Company, Contamac, ZEISS Group, SynergEyes, Menicon Co., Ltd., Gelflex, Orion Vision Group, Solotica, medios, SEED CO. LTD, and others.
Intraocular Lens Market Insights, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the market trends, market drivers, market barriers, and key intraocular lens companies, including Johnson & Johnson, HOYA GROUP, Alcon Inc., Carl Zeiss Meditec AG, Bausch & Lomb, Rayner Intraocular Lenses Limited, Eyekon Medical Inc., Lenstec, Inc., HumanOptics AG, STAAR Surgical Company, Aurolab, PhyIOL S.A., Care Group, Tekia Inc., Omni Lens Pvt Ltd, MORCHER® GmbH, Ophtec BV, SIFI S.p.A., Excellent Hi-Care Pvt. Ltd., Hanita Lenses, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.

Contact Us Shruti Thakur info@delveinsight.com +14699457679
© Copyright Globe Newswire, Inc. All rights reserved. The information contained in this news report may not be published, broadcast or otherwise distributed without the prior written authority of Globe Newswire, Inc.


